ANB033 — Eosinophilic Esophagitis
Phase 1b cohort in eosinophilic esophagitis initiated; top-line Phase 1b data in 2027
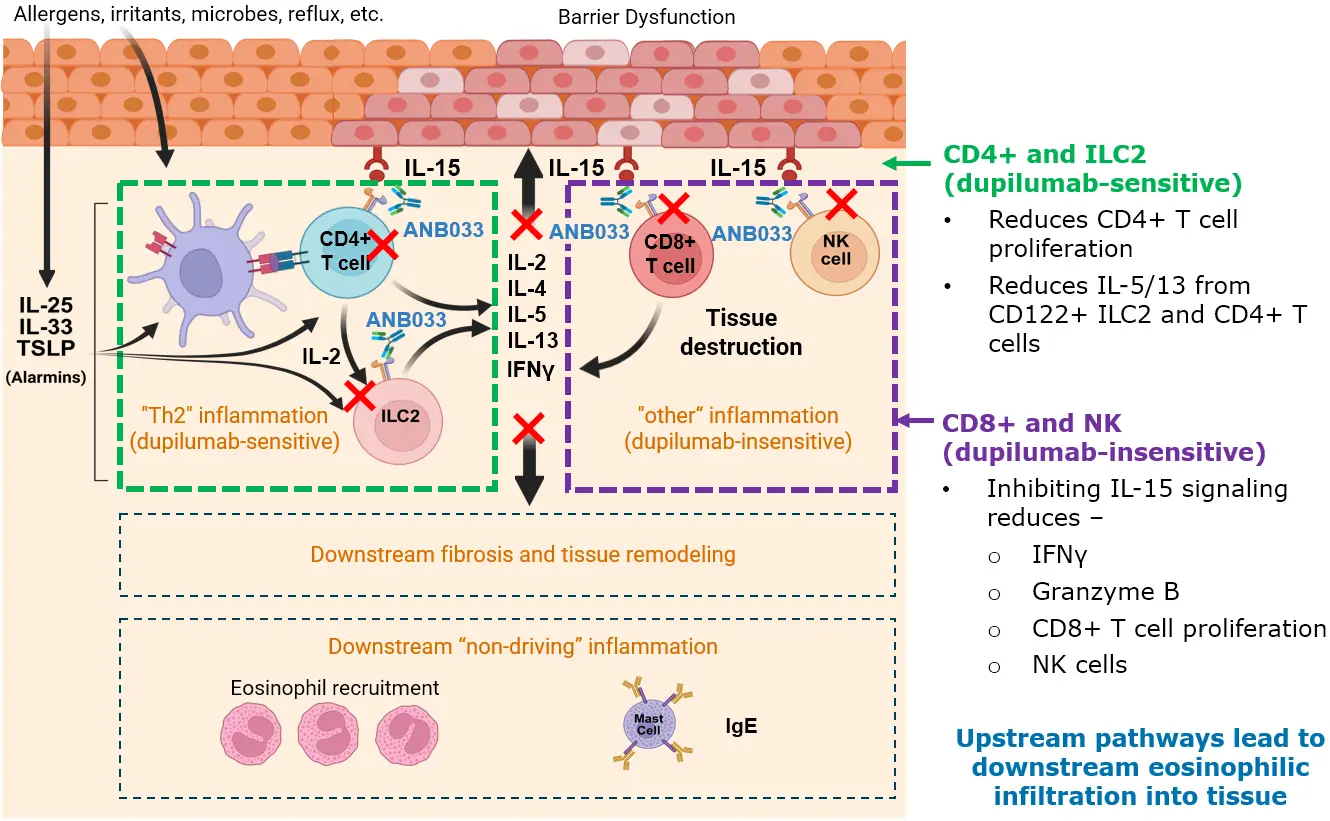
Similar to CeD, ANB033 targets multiple drivers of EoE biology addressing both dupilumab sensitive and insensitive pathways
Adapted from Discepolo et. al. Gastroenterology. 2024; 167:90-103.
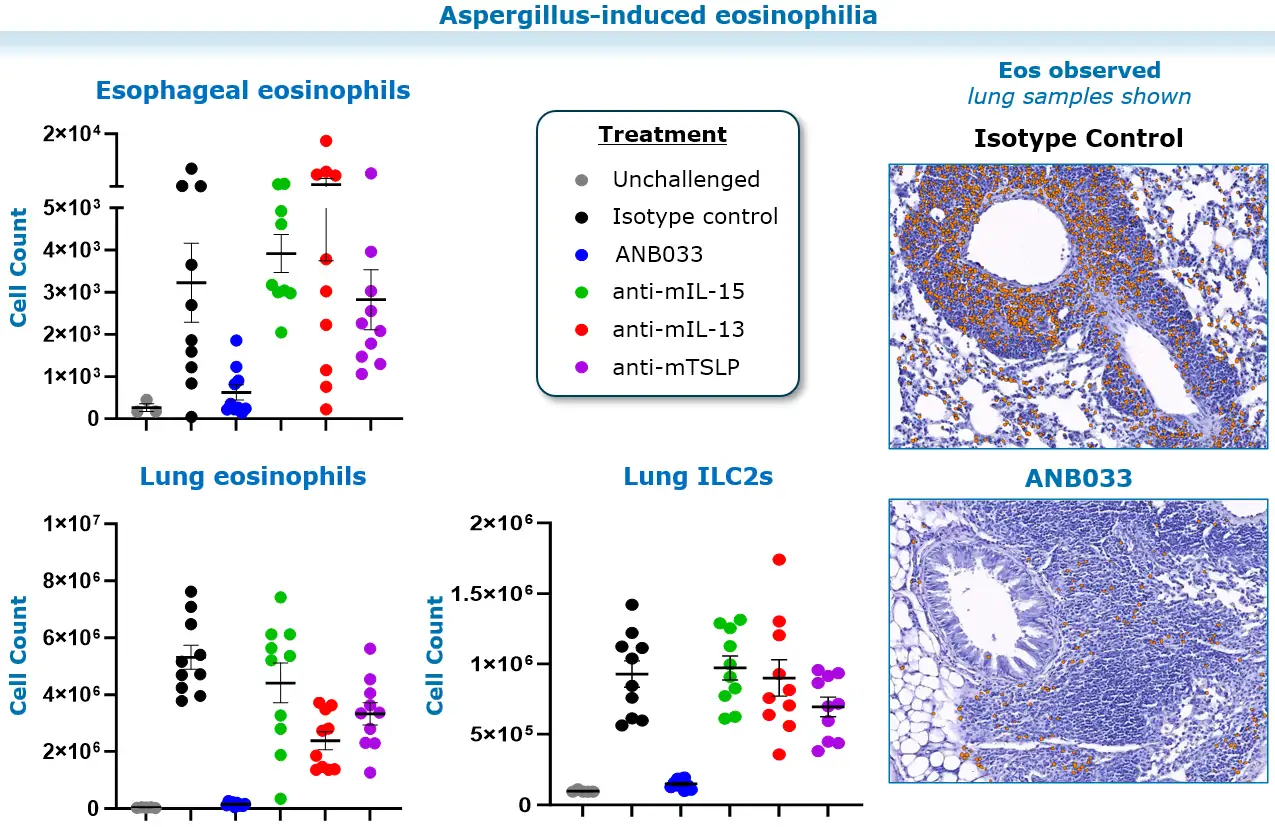
Model of eosinophilic inflammation: Balb/c mice were challenged intranasally with Aspergillus fumigatus TIW for 3 weeks. The treatment regimen includes a saline, isotype control, and ANB033 surrogate antibody (anti-mouse CD122 antibody with similar binding epitope and affinity to ANB033, administered at 10 mg/kg BIW for 3 weeks. Tissues were stained with H&E for histopathology assessment.
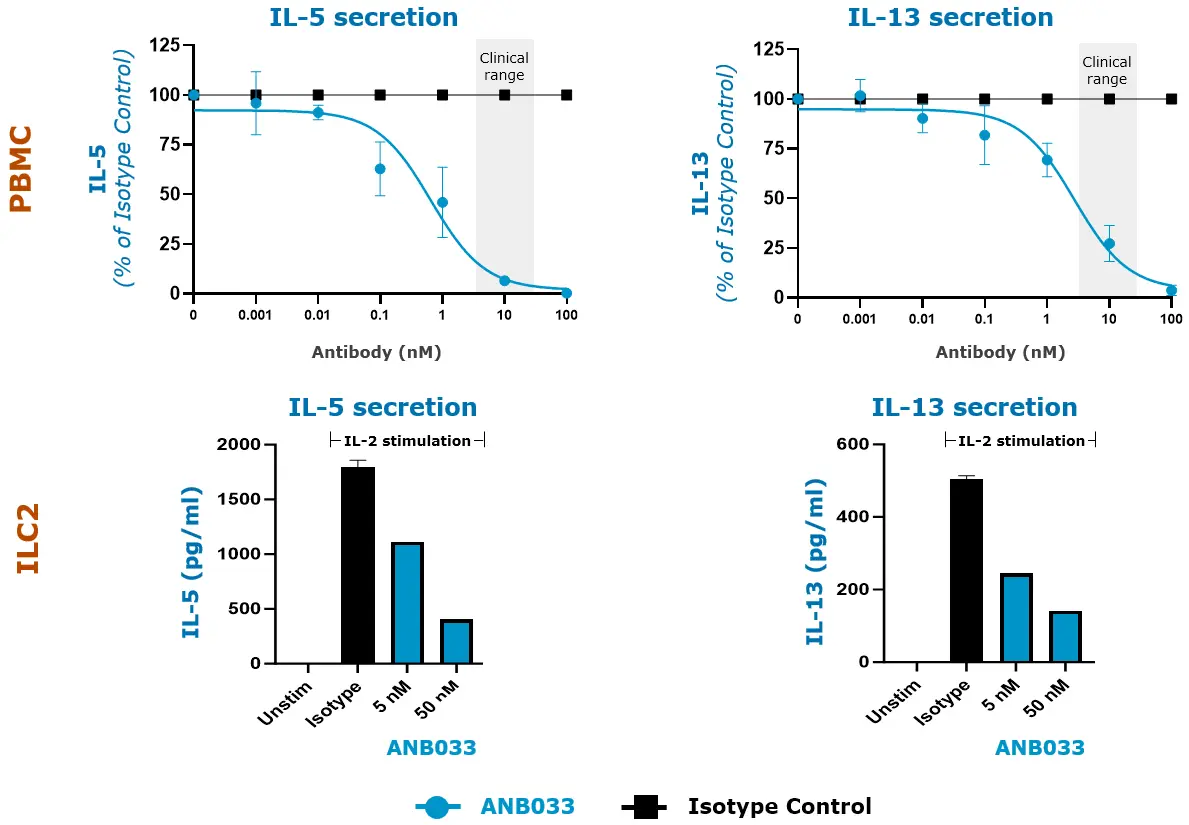
Top Panel: Human healthy PBMC were activated by anti-CD3/CD28 for 3 days; n=4 donors shown.
Bottom Panel: Purified human whole blood-derived ILC2 maintained in IL-33 were stimulated with IL-2 for 3 days; 1 of 6 similar representative donors shown.
To access scientific publications on this topic, please click here.